Deregulated expression of c-mos oncogene in non-small
cell lung carcinomas. Relationship with p53 protein status, genomic
instability and tumor kinetics
Gorgoulis G.V.1, Zacharatos P.1, Evagelou
K.1, Sergentanis T.1, Mariatos G.1,
Kotsinas A.1, Foukas P.1, Economopoulou C.1,
Liontos M.1, Giannoukakos D.2, Trigidou R.3,
Karameris A.4, Kanavaros P.5, Barbatis K.6,
Kittas C.1
1Department of Histology and Embryology, School of
Medicine, University of Athens
2Laboratory of Radioisotopes, NCSR "Demokritos", Athens
3Department of Pathology, "Sotiria" Hospital, Athens
4Department of Pathology, "NIMITS" Army Hospital, Athens
5Department of Histology and Embryology, School of Medicine,
University of Thessalia
6Department of Pathology, "Red-Cross"
Hospital, Athens
Aim: Little is known about the status of the mitogen-activating
protein kinase (MAPK) pathways in lung cancer. The product of
the c-mos proto-oncogene is one of the key molecules taking part
in these pathways. In vitro investigations have shown that it
is involved in oocyte maturation, whereas in somatic cells it
has opposing effects on the cell cycle suggesting that this proto-oncogene
may represent an important determinant of aberrant cell function
(genomic instability and altered kinetics). A recent study suggests
that these effects may be p53-dependent. In view of the proposed
link between c-mos and p53, we examined in non-small cell lung
carcinomas (NSCLCs): i) the status of c-mos, ii) its relationship
to genomic instability (aneuploidy) and two kinetic parameters
of the tumors, proliferation (PI) and apoptotic indexes (AI),
and iii) its association with p53 alterations and their concomitant
relationship with the above parameters.
Material and Methods: The study was performed on 56 NSCLCs.
The proliferation index (PI), the apoptotic index (AI) and ploidy
status were evaluated by Ki-67 immunohistochemistry, TUNEL assay
and Feulgen-DNA staining and image analysis, respectively. C-mos
and p53 in-situ protein levels were assessed by immunohistochemistry.
Moreover, c-mos and p53 status were examined by LOH, mutation
analysis and RT-PCR. In addition, any possible correlation among
the examined parameters was performed by statistical analysis.
Results and Conclusions: C-mos overexpression, as assessed
by immunohistochemistry and mRNA analysis, was found in 27% of
the tumors. Expression was higher in advanced stages (II&III vs
I, P=0.018). Since c-mos gene amplification was not detected,
its deregulated expression may be due to increased transcription.
The majority of c-mos positive [c-mos(P)] cases was associated
with aneuploidy. Sequencing showed two silent mutations and one
missense (R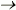 L)
at codon 22, located in a region critical for c-mos stability.
In contrast to the findings of some in vitro studies, c-mos overexpression
was found to be accompanied with low AI score tumors, implying
that induction of apoptosis may have been defective. Indeed, in
these tumors p53 alterations were found. The carcinomas with concomitant
alterations of c-mos and p53 [c-mos(P)/p53(P)] had significantly
lower AI values (P<0.001) and were more frequently associated
with aneuploidy (P=0.015) than the c-mos(N)/ p53(N) tumors, but
not the c-mos(N)/p53(P) tumors. The former suggests that p53 status
is the main determinant of ploidy status and apoptosis in our
series. This finding reveals once again the pivotal role of wild-type
(wt) p53 in preventing oncogene-mediated activation. L)
at codon 22, located in a region critical for c-mos stability.
In contrast to the findings of some in vitro studies, c-mos overexpression
was found to be accompanied with low AI score tumors, implying
that induction of apoptosis may have been defective. Indeed, in
these tumors p53 alterations were found. The carcinomas with concomitant
alterations of c-mos and p53 [c-mos(P)/p53(P)] had significantly
lower AI values (P<0.001) and were more frequently associated
with aneuploidy (P=0.015) than the c-mos(N)/ p53(N) tumors, but
not the c-mos(N)/p53(P) tumors. The former suggests that p53 status
is the main determinant of ploidy status and apoptosis in our
series. This finding reveals once again the pivotal role of wild-type
(wt) p53 in preventing oncogene-mediated activation.
Key words: mos, p53, proliferation, apoptosis, ploidy.
|